Зоологический журнал, 2021, T. 100, № 3, стр. 243-255
Morphological polymorphism of Longinebela tubulosa (Arcellinida, Hyalospheniformes) from East Herzegovina
S. D. Luketa *
University of Novi Sad, Faculty of Sciences, Department of Biology and Ecology
21000 Novi Sad, Trg Dositeja Obradovića 2, Serbia
* E-mail: stefanluketa@yahoo.com
Поступила в редакцию 18.12.2019
После доработки 19.01.2020
Принята к публикации 30.01.2020
Аннотация
The genus Longinebela includes five testate amoeba species characterized by large, elongated-pyriform shells. The lateral margins tapering towards the aperture create a somewhat distinct neck. Longinebela tubulosa is the type species of the genus and has a cosmopolitan distribution, yet being very poorly studied in terms of morphological variability. In this paper, the morphological variability of L. tubulosa is presented, based on 2630 specimens from a Sphagnum-dwelling population from East Herzegovina. The minimal variation coefficient (5.49%) was observed for the shell width/shell length ratio, while the maximal variability (16.38%) was observed for the area of the optical section. The results of a size frequency distribution analysis of shell length and shell width indicate that this population possesses continuous polymorphism. Environmental and biological causes that may lead to polymorphism have been discussed and the need for further study of the molecular diversity, ecology and life cycle of this population has been outlined.
Species delimitation of testate amoebae possesses great difficulties because of the limited number of good morphological characters in some species groups. The problem is further complicated because some species are polymorphic. Polymorphism is simultaneous presence of two or more phenotypes in a population or interrupted diversity of forms on the common genetic base. In the cases of continuous polymorphism, it is not possible to distinguish two or more clearly defined morphs within a population; this situation is relatively common among testate amoebae (Schönborn, Peschke, 1988; Schönborn, 1992; Wanner, 1999; Bobrov, Mazei, 2004; Golemansky, Todorov, 2006; Todorov, 2010). In the cases of discontinuous polymorphism, it is possible to distinguish clearly defined morphs within population. This type of polymorphism was registered in several cases among testate amoebae (Schönborn, 1992; Foissner, Korganova, 1995; Zapata, Fernández, 2008; Bobrov et al., 2013; Luketa, 2016a, 2017; Todorov et al., 2018). Both molecular and morphological taxonomists often overlook or minimize polymorphism by sampling only a single or few specimens per species.
Majority of testate amoebae with lobopodia are included into order Arcellinida which comprising about 700 nominal species, but the major part of these species needs comprehensive taxonomic revision. Lahr et al. (2019) based on a well-resolved phylogenomic reconstruction defined the infraorder Hyalospheniformes. With more than 200 described taxa distributed over all continents, this group is both highly variable and one of the largest testate amoebae groups. The precise taxonomy of the hyalospheniids is still poorly understood with incorrect descriptions being widespread, so today only about 100 species are accepted. Many of hyalospheniids are clearly identifiable by their large size and by the characteristic shell shape, so traditional taxonomic methods are still used today to describe new morphospecies (Todorov, 2010; Bobrov, Kosakyan, 2015; Todorov et al., 2018). The increasing use of molecular methods in taxonomic studies showing the presence of a large pseudocryptic diversity in many broadly defined morphospecies (Kosakyan et al., 2012, 2013, 2016; Singer et al., 2015; Qin et al., 2016). Molecular studies often underestimates infraspecific variability on intrapopulation and interpopulation levels, so several recently published studies have been devoted to the morphological variability of earlier described species (Török, 2001; Todorov, 2002; Todorov et al., 2010; Luketa, 2015, 2015a, 2016, 2017, 2017a, 2017b, 2017c, 2017d; Nicholls, 2015).
The genus Longinebela comprises five species (L. ampulla, L. tubulosa, L. meisterfeldi, L. speciosa and L. golemanskyi) characterized by elongated-pyriform shell shape in broad lateral view, laterally compressed. The lateral margins tapering toward the aperture, creating a somehow distinct neck; small lateral pores are usually present, but hard to observe. The shell is yellowish brown, composed usually of circular, oval or quadrangular plates, probably recycled from predated small testate amoebae (e.g. members of the genera Trinema, Corythion and Quadrulella); sometimes amoebae possess shells composed of voluminous, globular or rarely elongated, rounded and hollow plates, usually with large parts between them interspersed with small beads of organic cement. Type species ‒ L. tubulosa ‒ is widespread and common, but very poorly studied in terms of morphological variability. Although many authors have recorded basic morphometric data for different populations (Penard, 1890, 1905; Cash, Hopkinson, 1909; Wailes, 1912; Grandori, 1934; Deflandre, 1936; Jung, 1936; Van Oye, 1941, 1956; Hoogenraad, de Groot 1952; Štӗpánek, 1963; Ogden, Hedley, 1980; Badewitz, 2002; Kosakyan et al., 2016; Todorov, Bankov, 2019), no detailed morphometric analysis of any population of this species has been performed so far. That kind of studies are necessary to establish possible presence of pseudocryptic taxa hidden within this highly variable morphospecies. This possibility is further supported by the fact that all other species of this genus have limited geographical distribution. Smith et al. (2007) concluded that L. speciosa is a good candidate for Northern endemism, but Zapata et al. (2008) recorded this species in Chile. It seems that L. speciosa has a wide geographical distribution, but with infrequent findings and only in populations with low number of specimens. The morphological variability of this species has been poorly studied and there are only basic morphometric data available for it (Deflandre, 1936; Ogden, 1984; Todorov, Bankov, 2019). Recently, three species (L. golemanskyi, L. meisterfeldi, and L. ampulla) that are thought to have very limited distribution have been described (Todorov, 2010; Kosakyan et al., 2012; Todorov et al., 2018).
I performed detailed morphometric investigation of the shell variability of Longinebela tubulosa based on a sphagnicolous population from East Herzegovina. The aim of this study was to test, by morphometric analyses, whether this population is monomorphic or polymorphic and to provide the basis for further investigation of ecomorphological variation and molecular diversity in this hyalospheniid testate amoeba.
MATERIAL AND METHODS
The material for the present study was extracted from several Sphagnum species collected in the peatland located in the Alagovac Lake region (43°17′44.8″ N, 18°07′31.9″ E, ca. 850 m a. s. l.), municipality Nevesinje, East Herzegovina on 18 April 2014, 19 August 2014, 11 May 2016, and 24 July 2016. Morphological characters and morphometric variables were studied using a light microscope Zeiss Axio Imager A1. Images were captured using an AxioCam MRc5 (Zeiss) digital camera. Measurements were conducted in the program AxioVision 4.9.1. The following measurements were taken for the studied shells: shell length, shell width, aperture width, and area of the optical section (area enclosed by the outline of the shell). The following descriptive statistics were calculated: extreme values (minimum and maximum), median, arithmetic mean, standard error of the arithmetic mean, standard deviation, coefficient of variation (in percentage), skewness and kurtosis. Statistical analysis was conducted using the program Statistica 13.0.
RESULTS
Description. The shell is elongated pyriform in broad lateral view, with sides tapering evenly and gradually from rounded aboral region towards the aperture, forming a somehow short distinct neck. The shell is laterally compressed and have oval transverse section. The lateral keel beginning at the base of the neck and extending all around. From the lateral view the shell have a pointed posterior end of the fundus. Two small lateral pores are usually present on the first one third of lateral margins. The shell is transparent, yellow to brown, composed mainly of small overlapping plates of different shape (oval, circular and/or square), arranged haphazardly and embedded in thick layer of organic cement. The aperture is oval, slightly convex, bordered by a thin organic lip. Figure 1 shows light micrographs of specimens from the studied population.
Fig. 1.
Light micrographs of Longinebela tubulosa: broad lateral view of different specimens from the Alagovac Lake region, East Herzegovina. Scale bars 50 μm.

Morphometry. Morphometric characters of 2630 specimens of L. tubulosa from the Alagovac Lake region were measured on April 2014, August 2014, May 2016, and July 2016. There are no significant differences between the median values of the characters in different periods (Fig. 2), which indicates stability of morphometric features. The largest difference was observed for the length and width of the shell between spring and summer specimens. Namely, spring specimens are slightly shorter and narrower than summer ones. These differences are not large, so the data for all periods are merged together (Table 1). Coefficient of variation was moderate for area of the optical section (16.38%), while other measured parameters were characterized by low variability (from 5.49 to 8.82%). For basic characters, the minimal variability was observed for aperture width (5.64%), while the maximal variation coefficient was observed for area of the optical section (16.38%). For ratio characters, the minimal variability was observed for shell width/shell length ratio (5.49%), while the maximal variation coefficient was observed for aperture width/shell length ratio (8.53%).
Fig. 2.
Inter-seasonal morphological variability of Longinebela tubulosa based on 2630 specimens from the Alagovac Lake region, East Herzegovina. Box plots show the variability of the shell length (A), shell width (B), aperture width (C), shell width/shell length ratio (D), aperture width/shell length ratio (E), and aperture width/shell width ratio (F). Legend: a ‒ 413 specimens collected on 18 April 2014, b ‒ 1146 specimens collected on 19 August 2014, c ‒ 751 specimens collected on 11 May 2016, d ‒ 320 specimens collected on 24 July 2016, square ‒ median, box ‒ 25%‒75%, whisker ‒ non-outlier range.
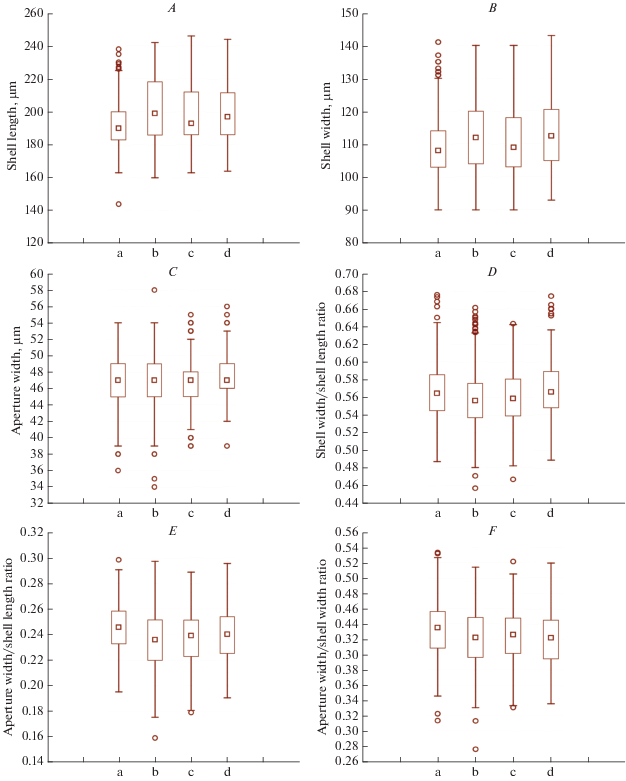
Table 1.
Morphometric characterization of Longinebela tubulosa from East Herzegovina based on 2630 specimens (measurements in μm)
| Characters | Min | Max | M | x | SE | SD | CV | Sk | Ku |
|---|---|---|---|---|---|---|---|---|---|
| Shell length | 144 | 246 | 194 | 198.70 | 0.33 | 17.06 | 8.59 | 0.347 | ‒0.916 |
| Shell width | 90 | 143 | 110 | 111.40 | 0.19 | 9.83 | 8.82 | 0.332 | ‒0.699 |
| Aperture width | 34 | 58 | 47 | 47.01 | 0.05 | 2.65 | 5.64 | ‒0.172 | 0.623 |
| Area of the optical section | 10 420 | 26 217 | 16 081 | 16 823 | 53.72 | 2755 | 16.38 | 0.405 | ‒0.914 |
| Shell width/shell length | 0.46 | 0.68 | 0.56 | 0.56 | 0.00 | 0.03 | 5.49 | 0.328 | 0.228 |
| Aperture width/shell length | 0.16 | 0.30 | 0.24 | 0.24 | 0.00 | 0.02 | 8.53 | ‒0.115 | ‒0.420 |
| Aperture width/shell width | 0.28 | 0.53 | 0.43 | 0.42 | 0.00 | 0.03 | 8.19 | ‒0.100 | ‒0.289 |
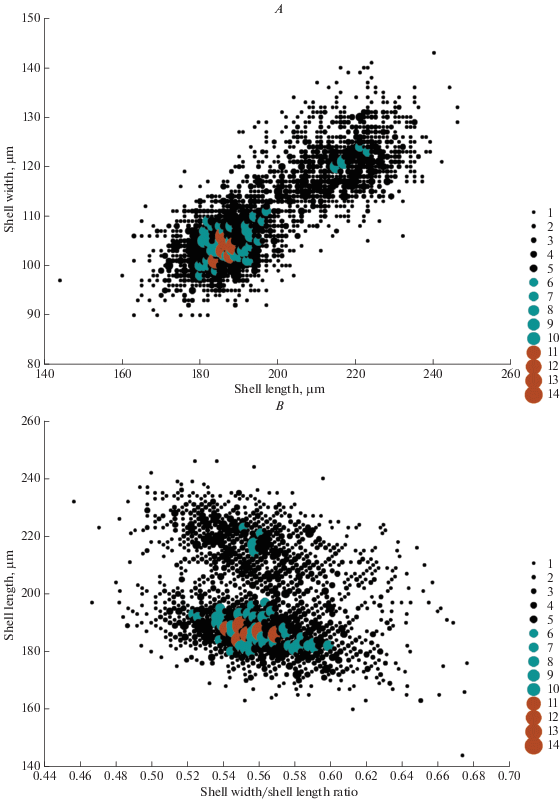
The most frequent shell length (186, 188 and 189 μm) was registered in 85 specimens (Fig. 3A); the most frequent shell width (103 μm) was registered in 120 specimens (Fig. 3B), and the most frequent aperture width (47 μm) was registered in 415 specimens (Fig. 3C). Analysis of the size frequency distribution of shell length and shell width indicates that this population is characterized by continuous polymorphism. All measured specimens had shell length between 144 and 246 μm. In this case, 66.27% of all specimens had shell length between 178 and 212 μm, whereas only 7.34% were smaller than 178 μm and 26.29% were larger than 212 μm. The frequency analysis of shell width shows a slightly different distribution pattern. All measured specimens had shell width between 90 and 143 μm, but in this case 49.39% of all specimens had shell width of 108‒125%, whereas 41.83% had shell narrower than 108 μm and only 8.78% had shell wider than 125 μm. Analysis of the size frequency distribution of aperture width indicates that this population is size-monomorphic. Aperture width ranged from 34 to 58 μm. However, 89.43% of all measured specimens had aperture width of 42‒50 μm, whereas only 1.79% had aperture narrower than 42 μm and only 8.78% had aperture wider than 50 μm. Figures 3D‒3F show bag plot analyses of the correlation between shell length, shell width and aperture width. The frequency scatter plot analyzes of the correlation between shell length and shell width (Fig. 4A), and shell width/shell length ratio and shell length (Fig. 4B) clearly show polymorphism within the studied population.
Fig. 3.
Morphological variability of Longinebela tubulosa based on 2630 specimens from the Alagovac Lake region, East Herzegovina. Histograms show the size frequency distribution of the shell length (A), shell width (B), and aperture width (C); bag plots show the correlation between shell length and shell width (D), aperture width and shell length (E), and aperture width and shell width (F). Legend for bag plots: depth median ◆, characters on Y axes ⚫, outliers ◼.
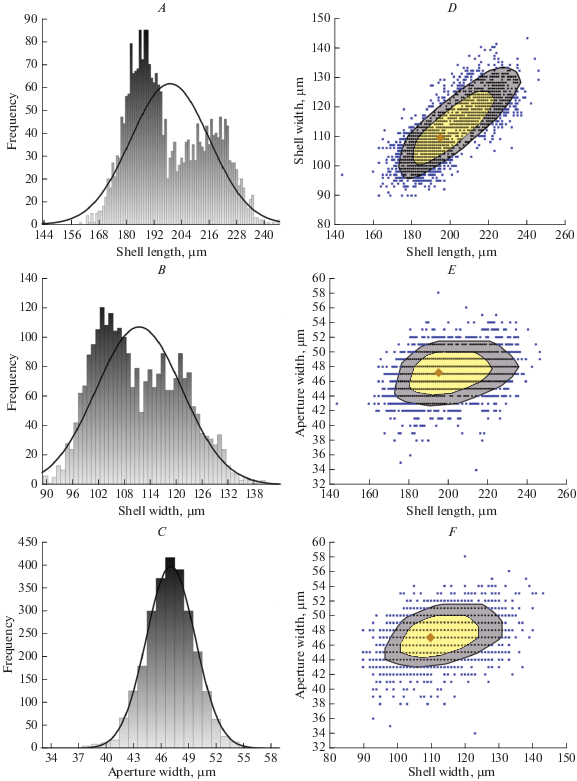
Fig. 4.
Frequency scatter plots show the correlation between shell width and shell length (A), and shell width/shell length ratio and shell length (B) of 2630 specimens of Longinebela tubulosa from the Alagovac Lake region, East Herzegovina.
The negative skewness values for aperture width (‒0.172), aperture width/shell length ratio (‒0.115), and aperture width/shell width ratio (‒0.100) suggest an asymmetrical distribution with a long tail toward lower values. Since the negative values are not clearly different from zero, the asymmetry of size distribution of these variables were minimal. All other variables were characterized by moderate positive skewness values (0.328‒0.405). Low positive kurtosis value (0.228) was observed for shell width/shell length ratio, while high positive value (0.623) was registered for aperture width. These characters were characterized by sharper distribution than a standard Gaussian distribution. Other variables were found to have negative kurtosis values (between ‒0.916 and ‒0.289), indicating a distribution that is flatter than the standard Gaussian distribution. Moderate negative value (‒0.289) was observed for aperture width/shell width ratio, while high negative kurtosis values were registered for shell length (‒0.916), area of the optical section (‒0.914), shell width (‒0.699), and aperture width/shell length ratio (‒0.289).
DISCUSSION
In this study it has been shown that population of Longinebela tubulosa from East Herzegovina is very heterogeneous, indicating the possible presence of pseudocryptic diversity or polymorphism. It is possible to assume that L. tubulosa is a species complex and that certain cryptic and/or pseudocryptic species are endemic for certain geographical areas. Laminger (1973) in a population from Austria noticed two unusual specimens: one specimen has a significantly wider fundus (shell length 183 μm, shell width 137 μm, aperture width 43 μm), while the other specimen has a remarkably narrowed neck with widens at the aperture (shell length 291 μm, shell width 123 μm, aperture width 49 μm). Both forms were found in the hydrophilic mosses. Hoogenraad and de Groot (1952) found an extremely variable population in Sphagnum teres collected in the Kamchatka Peninsula (Russia). The shell length varied between 140 and 307 μm, while the shell width was between 80 and 150 μm. In morphometric terms, this population is the most variable among L. tubulosa populations analyzes so far, although it inhabits only single moss species. Laminger (1971) described L. tubulosa var. adami (as Nebela tubulosa var. adami) from Austria that differs from its type in that the lip is better developed and the shell is much smaller (shell length 101‒114 μm, shell width 52‒65 μm, and aperture width 18‒22 μm). Given the large and discontinuous differences in the size of the shell, it can be assumed that it is a separate species, but detailed studies of this taxon are needed to clarify its taxonomic status and position. Comparative morphometric data of L. tubulosa according to different authors are presented in Table 2.
Table 2.
Comparative morphometric data (minimum‒maximum) of Longinebela tubulosa according to different authors (all measurements in μm)
| Location | Shell length | Shell width | Aperture width | Reference |
|---|---|---|---|---|
| Germany | 220‒240 | 80‒90 | – | Penard, 1890 |
| Switzerland | 200‒215 | – | – | Penard, 1905 |
| Great Britain | 190‒220 | – | – | Cash, Hopkinson, 1909 |
| United States of America | 174‒196 | 74‒93 | 30‒35 | Wailes, 1912 |
| Italy | 190‒215 | – | – | Grandori, 1934 |
| – | 190‒215 | 80‒125 | 35‒63 | Deflandre, 1936 |
| Germany | 222‒234 | 120‒130 | 48‒57 | Jung, 1936 |
| Iceland | 215 | 121 | 55 | Van Oye, 1941 |
| Sakhalin Island, Russia | 183‒217 | 100‒130 | – | Hoogenraad, de Groot, 1952 |
| Kamchatka Peninsula, Russia | 140‒307 | 80‒150 | – | Hoogenraad, de Groot, 1952 |
| Venezuela | 190‒205 | 90‒101 | 30‒40 | Van Oye, 1956 |
| Congo | 188 | 98 | 38 | Štӗpánek, 1963 |
| Great Britain | 213‒264 | 120‒155 | 42‒54 | Ogden, Hedley, 1980 |
| Germany | 208‒226 | 104‒140 | – | Badewitz, 2002 |
| Bulgaria | 189‒282 | 92‒168 | 42‒68 | Todorov, Bankov, 2019 |
| East Herzegovina | 144‒246 | 90‒143 | 34‒58 | This study |
Molecular approaches on hyalospheniids opened new horizons in biogeography and ecology of testate amoebae. Namely, cryptic species showed non-overlapping geographic distribution areas (Heger et al., 2013), disproving the cosmopolitan hypothesis for hyalospheniids at least. Pseudocryptic and cryptic species also showed diverging ecological preferences for different microniches in Sphagnum peatlands (Singer et al., 2015, 2018). Singer et al. (2018) concluded that if cryptic species differ in their ecological optima and/or function, then potentially useful information for biomonitoring or paleoecological inference is lost by lumping. If the species can not be identified by morphological or morphometric characters then applications would only be limited to the living communities, so if they do not differ in ecology or function then lumping them in morphospecies seem to be a reasonable approach. Therefore, it is necessary to study the morphology, morphometry, biogeography and ecology of individual populations within the morphospecies before the final conclusions are made.
From the evolutionary point of view, a very interesting example is Difflugia tuberspinifera. It is an endemic species of East Asia and is common in freshwater lakes and reservoirs in China. Yu et al. (2014) during investigations of the protist diversity in Xiamen reservoirs (China) found abundant material of living specimens of Difflugia tuberspinifera, so they performed detailed morphometric analysis based on 1165 specimens. They registered about 45% specimens had no spines and concluded that within D. tuberspinifera there are two distinct morphotypes ‒ spiny and spineless types. Namely, their results indicated that on all metric variables but excluding spine number did not allow differentiation of spineless type from spiny type, so these two morphotypes either could be considered as two different morphotypes of the same species or two closely related species. It remained unclear if the absence of spines was genetically determined and if the presence or absence of spines is a valid taxonomical criterion for arcellinid testate amoebae in general or if is just on expression of phenotypic plasticity. However, Gomaa et al. (2015) based on molecular analysis showed that the two morphotypes are genetically distinct, but very closely related. Their phylogenetic data analyses based on the ITS sequences clearly showed that all inter-specific variations were greater than intra-specific variations, and a clear gap existed between both morphotypes. Also, the fact that the ITS sequences of the spinose morphs form a clade, whereas the spineless morphs do not, suggests that the spinose morphotypes might have evolved from spineless ancestors. The spinose morphotype has a wider distribution than the spineless morphotype. It is a remarkable fact if the spinose morphotype was derived from a spineless ancestor. However, high population densities of spinose morphotype were observed in many artificial reservoirs that were constructed after the Second World War, so this may indicate that the spinose morphotype has a competitive advantage in these reservoirs and is more adapted to local ecosystem conditions (Gomaa et al., 2015). Probably, it is a case of a rapid evolution of a new morphotype which has no time to suppress the typical morphotype due to its short existence. It is also possible that these two morphotypes have occupied different ecological niches and will continue to live in sympatry. In D. tuberspinifera a more comprehensive sampling and the analysis of additional genetic markers are necessary to clarify taxonomic status of two morphotypes and the biological relevance of existing difference. This example shows how complex and rapid may be evolutionary processes in testate amoebae, and that molecular analyzes are not sufficient to correctly determine the taxonomic status of some morphotypes.
Logares et al. (2007) investigated the dinoflagellate morphospecies Peridinium aciculiferum and Scrippsiella hangoei which show significant interspecific differences in the external morphology. Despite these phenotypic differences and the habitat segregation, molecular analyses showed identical ribosomal DNA sequences for both morphospecies. Geographical isolation most probably played a minor role in the divergence of these species, because Lake Erken (habitat of P. aciculiferum) has been connected to the Baltic Sea (habitat of S. hangoei) through a network of flowing waters throughout its history. Namely, gene flow should have been possible between the diverging freshwater and marine lineages, at least in the direction from the lake to the sea. A rapid morphological divergence between these species is the most parsimonious explanation for their identical rDNA sequences. Logares et al. (2007) proposed a scenario in which P. aciculiferum marine ancestors were landlocked or invaded newly formed lakes after the last ice age, and diverged rapidly as a result of a strong selection caused by the new ecological conditions found in freshwater environments. The strong genetic isolation between P. aciculiferum and S. hangoei was shown by AFLP (amplified fragment length polymorphism), and the absence of shared cob haplotypes indicate no recent interspecific gene flow. If a future molecular study show that two morphotypes of Longinebela tubulosa have identical rDNA sequences, then genetic isolation between them should be tested.
Morphological polymorphism can be caused by environmental and biological factors. Environmental factors can be grouped into abiotic (soil type, nitrogen content) and biotic (Sphagnum species, prey type).
Schönborn (1992) have been conducted study about morphological and morphometric variability of the soil-dwelling euglyphid testate amoebae Trinema complanatum and Euglypha laevis that possess adaptive morphs of a discontinuous polymorphism. These cases are good examples of a sympatric model of morphological differentiation of subpopulations without genetic divergence. He analyzed shell morphometry of T. complanatum specimens that inhabit spruce forest soils with two horizons ‒ a litter (needle) and underlying humus horizon. It is possible to distinguish four shell morphs: large-waisted, small-waisted, large-rounded, and small-rounded. Analysis has shown that the waisted forms were distributed mainly in the needle horizon, while the rounded forms were inhabited the underlying humus horizon. The shell width/shell length index of the rounded forms is higher than that of the waisted forms, so the rounded shells are stouter than the waisted shells. This could be interpreted in the context of adaptation of shells in soils for protection against evaporation. Namely, stout forms are better adapted to pores of the humus horizon than waisted shells, while waisted shells are better adapted to litter horizon because they can use the thin water film on the needles. The shell indentation of waisted morphs effects the origin of an adhesion ring, so when the films dry up, the shells adhere to the needle surface and the adhesive ring closes the aperture. Schönborn (1992) also noted that in an adjoining Sphagnum plot only the waisted morphs has been found and concluded that it is because the water film on Sphagnum leaves may be similar to that of spruce needles. The small-rounded specimens can colonize compacted soils with small pore capacity, so it is probably they live in the narrower pores rather than large specimens. Similar case is polymorphism of Euglypha laevis because in soils it lives in both the litter and the humus horizon. Schönborn (1992) distinguished large and small morphs in the soil. Narrow and wide shells were found in small and large morphs, but they were not referred to horizons. Namely, narrow and wide forms were interpreted as individual varieties without ecological significance. The large morph was occur in Sphagnum due to the similar microhabitat structure on Sphagnum leaves and needle surfaces, but the size spectrum in Sphagnum is split into three peaks indicating unknown microhabitat structures. Apertures were mostly round, but there were also specimens with oval apertures which were found only in the large morph confined on the litter horizon.
Singer et al. (2018) concluded that irrespective of any other biotic interaction, the distribution of Nebela species was highly correlated with nitrogen content. Nutrient deficiency is common in Sphagnum-dominated peatlands (especially in hummocks) and shape prokaryotic communities, so it is likely that the especially low availability of nitrogen in hummocks acts as an environmental filter influencing species composition within hyalospheniid testate amoebae. For the community of testate amoebae, water table depth and pH are usually the two main gradients explaining community composition, but these abiotic factors not correlate with species composition within the genus Nebela (Singer et al., 2018). These findings suggest that species differ in their respective ecological preferences and that these factors may structure soil testate amoebae communities at biogeographical and microhabitat spatial scales. The ecological differences within the peatland were not taken into account in the study presented in this paper. Therefore, it is necessary to carry out an ecological study to examine the relationship of the morphotypes registered in this study to the nitrogen content of individual parts of the peatland.
Community patterns of soil microorganisms including testate amoebae may be directly or indirectly influenced by plants, especially Sphagnum mosses strongly influence key factors such as pH, nutrient and cation concentrations and water content (Clymo, 1973; Van Breemen, 1995; Lindo, Gonzalez, 2010). Todorov et al. (2018) presented the scatter plot of shell length versus shell width in population of Gibbocarina penardiana based on 100 specimens extracted from wet moss Sphagnum flexuosum collected on Stara planina Mountains (Bulgaria). This scatter plot illustrates that presented population of G. penardiana possesses discontinuous polymorphism, so two forms can be distinguished: small (shell lengrh varies between 127 and 157 μm) and large (shell length varies between 159 and 180 μm). Specimens of the large form correspond to the typical G. penardiana and those of the small form are closer to the infraspecific taxon N. penardiana var. minor, described from Africa (Gauthier-Lièvre, 1957). These results are important because show that polymorphism within the species is not always caused by moss species.
Testate amoebae are considered microbial top-predators, so they are likely to contribute to shaping microbial community structure. Also, the absence of certain prey organisms may influence their community composition (Gilbert et al., 2003). If different microhabitats are inhabited by different prey in same locality, it is possible that prey type influence the difference in body size of testate amoebae from different microhabitats. Shell length and aperture width are linked to feeding habit of testate amoebae and indicate the trophic position of each species with a low aperture width/shell length ratio (<0.18) were considered as having a low trophic position (i.e. primarily bacterivores and algivores), while species with a high one (>0.18) as having a high trophic position (i.e. primarily predators of other protists and micrometazoans) in the microbial food web (Jassey et al., 2013). Jassey et al. (2014) have shown that testate amoebae can be separated into two main groups ‒ large and small taxa ‒ based on their trophic position. According to this approach, species with a small shell length (<60 μm) and small aperture width (<15 μm) are usually grazers (i.e. species with a low trophic level) feeding on bacteria and fungi, while species with a large shell length (>60 μm) and large aperture width (>15 μm) are usually primarily predators (i.e. species with a high trophic level) feeding on other protists and micrometazoans. Both of these approaches indicate a positive correlation between the aperture size and the size of the prey on which testate amoeba species can predate. If we start from the assumption that there are two taxa within analyzed population of Longinebela tubulosa that consuming prey items of different size, a polymorphism in the size frequency distribution of the aperture width can be expected. Given that this population has a distinctly monomorphic size frequency distribution of aperture width, it can be reasonably assumed that differentiation is not caused by prey types.
The causes of morphological differentiation within a population may be of biological nature, i.e. related to reproduction. In this case, morphological differences may be caused by sexual dimorphism or plastogamy. The nature of these phenomena is much less studied than environmental factors. Oliverio et al. (2014, 2015) have suggested that Hyalosphenia papilio and H. elegans represent two different mating types of the same species. This phenomenon has not been well studied, and a future study on the reproduction of Longinebela tubulosa will be necessary to clarify whether the dimorphism of the studied population from East Herzegovina is caused by the presence of different mating types.
Plastogamy is the fusion of the cytoplasm of two or more cells without the fusion of their nuclei. Some amoeboid protist groups, especially Foraminifera and Gromiida, contain species that undergo plastogamy at particular times during their life cycles (Myers, 1935, 1940; Grell, 1958, 1958a; Weber, 1965; Arnold, 1966; Lipps, Erskian, 1969). Lipps and Erskian (1969) observed the foraminiferan Glabratella ornatissima and noted a dimorphism: one group consisted of larger, more conical and more thickly calcified specimens whereas another group consisted of smaller, lower specimens with flatter apertural side. Members of this smaller group participated in plastogamy. Reukauf (1912) described life cycle of testate amoeba Scutiglypha acanthophora and noted that after copulation of two specimens they fusing protoplasts and sharing the reserve plates accumulated on both sides, and that way produce a significantly larger specimen than either of the two copulants. This large specimen produces a cyst, so the encysted specimens are always characterized by striking size in comparison to the normal specimens. The two specimens involved in copulation are usually of unequal size, so it is assumed that this is a case of sexual dimorphism. For the purpose of copulation, the two specimens interconnect via apertures, as in the case of normal conjugation. However, they later to oppose each other at a certain angle. Now, the two protoplasts are fusing to form a significantly larger shell from common reserve plates. Wailes (1912) observed an abnormally large specimen of Longinebela tubulosa (shell length 300 μm, shell width 150 μm, aperture width 40 μm) at Lakehurst, New Jersey. He assumed that it may have been a case of plastogamy or double encystment. A detailed study of the life cycle of L. tubulosa population from East Herzegovina is needed to determine whether plastogamy is present and whether it is the cause of registered polymorphism.
CONCLUSIONS
The population of Longinebela tubulosa from the Alagovac Lake region is polymorphic, but size-classes are not clearly separated ‒ so, this population is an example of continuous polymorphism. Morphological polymorphism can be caused by environmental and biological factors. Environmental factors can be grouped into abiotic and biotic. Probably the most significant abiotic factor that could cause polymorphism within the studied population is nitrogen content, and future ecological studies of this population should focus primarily on this factor. Of the biotic factors, particular attention should be paid to the moss species on which specimens live, since the studied peatland contains several Sphagnum species. Biological factors are related to reproduction, but these phenomena are poorly understood in testate amoebae. The detailed studies on the biology of these protists are needed to speak more specifically about role of biological factors in evolutionary processes. Nevertheless, it can be stated that sexual dimorphism and plastogamy represent two phenomena that can cause polymorphism within a population. The existence of polymorphic species has profound implications for the study of hyalospheniid molecular diversity, ecology, reproduction, nutrition and biogeography.
Список литературы
Arnold Z.M., 1966. Observations on the sexual generation of Gromia oviformis Dujardin // Journal of Protozoology. V. 13. P. 23‒27.
Badewitz H.-J., 2002. Testaceen-Taxozönose (Rhizopoda, Testacea) mit hoher Artendiversität in einem kleinen Quellgewässer des Oberharzes // Lauterbornia. V. 44. P. 1‒28.
Bobrov A., Kosakyan A., 2015. A new species from mountain forest soils in Japan: Porosia paracarinata sp. nov., and taxonomic concept of the genus Porosia Jung, 1942 // Acta Protozoologica. V. 54. P. 289‒294.
Bobrov A., Krasilnikov P., Gracía-Calderón N.E., 2013. Biogeography of testate amoebae in the soils of Mexico // Biodiversity and Conservation. V. 22. P. 2837‒2855.
Bobrov A., Mazei Yu., 2004. Morphological variability of testate amoebae (Rhizopoda: Testacealobosea: Testaceafilosea) in natural populations // Acta Protozoologica. V. 43. P. 133‒146.
Cash J., Hopkinson J., 1909. The British freshwater Rhizopoda and Heliozoa. Volume II: Rhizopoda, part II. London: Ray Society. 166 p.
Clymo R.S., 1973. The growth of Sphagnum: some effects of the environment // Journal of Ecology. V. 61. P. 849‒ 869.
Deflandre G., 1936. Etude monographique sur le genre Nebela Leidy (Rhizopoda ‒ Testacea) // Annales de protistologie. V. 5. P. 201–322.
Foissner W., Korganova G.A., 1995. Redescription of three testate amoebae (Protozoa, Rhizopoda) from a Caucasian soil: Centropyxis plagiostoma Bonnet & Thomas, Cyclopyxis kahli (Deflandre) and C. intermedia Kufferath // Archiv für Protistenkunde. V. 146. P. 13‒28.
Gauthier-Lièvre L., 1957. Additions aux Nebela d’Afrique // Bulletin de la Société d’Histoire Naturelle de l’Afrique du Nord. V. 48. P. 494‒523.
Gilbert D., Mitchell E.A.D., Amblard C., Bourdier G., Francez A.J., 2003. Population dynamics and food preferences of the testate amoeba Nebela tincta major-bohemica-collaris complex (Protozoa) in a Sphagnum peatland // Acta Protozoologica. V. 42. P. 99‒104.
Golemansky V., Todorov M., 2006. New data to the shell ultrastructure and the biometry of the marine interstitial testate amoebae (Rhizopoda: Testaceafilosia) // Acta Protozoologica. V. 45. P. 301‒312.
Gomaa F., Yang J., Mitchell E.A.D., Zhang W.-J., Yu Z., Todorov M., Lara E., 2015. Morphological and molecular diversification of Asian endemic Difflugia tuberspinifera (Amoebozoa, Arcellinida): a case of fast morphological evolution in protists? // Protist. V. 166. P. 122‒ 130.
Grandori R.E.L., 1934. Studî sui Protozoi del terreno // Bollettino del Laboratorio di Zoologia Agraria e Bachicoltura del R. Instituto Superiore Agrario di Milano. V. 5. P. 1‒341.
Grell K.G., 1958. Untersuchungen über die Fortpflanzung und Sexualität der Foraminiferen, II. Rubratella intermedia // Archiv für Protistenkunde. V. 102. P. 291‒308.
Grell K.G., 1958a. Untersuchungen über die Fortpflanzung und Sexualität der Foraminiferen, III. Glabratella sulcata // Archiv für Protistenkunde. V. 102. P. 449‒472.
Heger T.J., Mitchell E.A.D., Leander B.S., 2013. Holarctic phylogeography of the testate amoeba Hyalosphenia papilio (Amoebozoa: Arcellinida) reveals extensive genetic diversity explained more by environment than dispersal limitation // Molecular Ecology. V. 22. P. 5172‒5184.
Hoogenraad H.R., de Groot A.A., 1952. Thekamöbe Moosrhizopoden aus Asien // Archiv für Hydrobiologie. V. 47. P. 263‒287.
Jassey V.E.J., Chiapusio G., Binet P., Buttler A., Laggoun-Défarge F., Delarue F., et al., 2013. Above- and belowground linkages in Sphagnum peatland: climate warming affects plant-microbial interactions // Global Change Biology. V. 19. P. 811‒823.
Jassey V.E.J., Lamentowicz Ł., Robroek B.J.M., Gąbka M., Rusińska A., Lamentowicz M., 2014. Plant functional diversity drives niche-size-structure of dominant microbial consumers along a poor to extremely rich fen gradient // Journal of Ecology. V. 102. P. 1150‒1162.
Jung W., 1936. Thekamöben ursprünglicher, lebender deutscher Hochmoore // Abhandlungen aus dem Landesmuseum der Provinz Westfalen, Museum für Naturkunde, Münster. V. 7. P. 1‒87.
Kosakyan A., Heger T.J., Leander B.S., Todorov M., Mitchell E.A.D., Lara E., 2012. COI barcoding of nebelid testate amoebae (Amoebozoa: Arcellinida): extensive cryptic diversity and redefinition of the Hyalospheniidae Schultze // Protist. V. 163. P. 415‒434.
Kosakyan A., Gomaa F., Mitchell E.A.D., Heger T.J., Lara E., 2013. Using DNA-barcoding for sorting out protist species complexes: a case study of the Nebela tincta‒collaris‒bohemica group (Amoebozoa; Arcellinida, Hyalospheniidae) // European Journal of Protistology. V. 183. P. 222‒237.
Kosakyan A., Lahr D.J.G., Mulot M., Meisterfeld R., Mitchell E.A.D., Lara E., 2016. Phylogenetic reconstruction based on COI reshuffles the taxonomy of hyalosphenid shelled (testate) amoebae and reveals the convoluted evolution of shell plate shapes // Cladistics. V. 32. P. 606‒623.
Lahr D.J.G., Kosakyan A., Lara E., Mitchell E.A.D., Morais L., Porfirio-Sousa A.L., et al., 2019. Phylogenomics and morphological reconstruction of Arcellinida testate amoebae highlight diversity of microbial eukaryotes in the Neoproterozoic // Current Biology. V. 29. P. 991‒ 1001.
Laminger H., 1971. Ein Beitrag zur Kenntnis der Thekamöbenfauna Österreichs (Protozoa, Rhizopoda testacea) // Zoologischer Anzeiger. V. 187. P. 372‒382.
Laminger H., 1973. Die Testaceenfauna (Protozoa, Rhizopoda) in der Umgebung von Obertauern (Salzburg) // Archiv für Protistenkunde. V. 115. P. 253‒270.
Lindo Z., Gonzalez A., 2010. The bryosphere: an integral and influential component of the Earth’s biosphere // Ecosystems. V. 13. P. 612‒627.
Lipps J.H., Erskian M.G., 1969. Plastogamy in Foraminifera: Glabratella ornatissima (Cushman) // Journal of Protozoology. V. 16. P. 422‒425.
Logares R., Rengefors K., Kremp A., Shalchian-Tabrizi K., Boltovskoy A., Tengs T., et al., 2007. Phenotypically different microalgal morphospecies with identical ribosomal DNA: a case of rapid adaptive evolution? // Microbial Ecology. V. 53 P. 549‒561.
Luketa S., 2015. Description of the family Padaungiellidae and morphological variability of Padaungiella lageniformis (Amoebozoides: Arcellinida) from the Vlasina Lake area, Serbia // Archives of Biological Sciences, Belgrade. V. 67. P. 1331‒1337.
Luketa S., 2015a. Morphological variability of two Quadrulella species (Arcellinida: Hyalospheniidae) from the Vlasina Lake region of Serbia // Biologia Serbica. V. 37. P. 22‒30.
Luketa S., 2016. Morphological variability of Porosia bigibbosa (Arcellinida: Hyalospheniidae) from East Herzegovina // Protistology. V. 10. P. 130‒137.
Luketa S., 2016a. First record of a size-trimorphic population among euglyphid testate amoebae // Protistology. V. 10. P. 138‒147.
Luketa S., 2017. Morphological variability of Padaungiella lageniformis (Arcellinida: Padaungiellidae) from the central part of the Balkan Peninsula // Protistology. V. 11. P. 20‒36.
Luketa S., 2017a. Morphological variability of Gibbocarina galeata and G. penardiana comb. nov. (Arcellinida: Hyalospheniidae) from East Herzegovina // Protistology. V. 11. P. 37‒47.
Luketa S., 2017b. Morphological variability of Nebela collaris s.s. (Arcellinida: Hyalospheniidae) from the Krečko Brdo Hill, East Herzegovina // Biologia Serbica. V. 39. P. 3‒8.
Luketa S., 2017c. Morphometrics of three hyalosphenid testate amoebae from the Velež Lake region, East Herzegovina // Protistology. V. 11. P. 196‒214.
Luketa S., 2017d. Taxonomy of Quadrulella longicollis and Q. symmetrica (Arcellinida: Hyalospheniidae) from the central part of the Balkan Peninsula // Protistology. V. 11. P. 215‒230.
Myers E.H., 1935. The life history of Patellina corrugate Williamson, a foraminifer // Bulletin of the Scripps Institution of Oceanography. V. 3. P. 355‒392.
Myers E.H., 1940. Observations on the origin and fate of flagellated gametes in multiple tests of Discorbis (Foraminifera) // Journal of the Marine Biological Association of the United Kingdom. V. 24. P. 201‒226.
Nicholls K.H., 2015. Nebela kivuense Gauthier-Lièvre et Thomas, 1961 (Amoebozoa, Arcellinida), missing for a half-century; found 11,500 km from “home” // Acta Protozoologica. V. 54. P. 283‒288.
Ogden C.G., 1984. Shell structure of some testate amoebae from Britain (Protozoa, Rhizopoda) // Journal of Natural History. V. 18. P. 341‒361.
Ogden C.G., Hedley R.H., 1980. An atlas of freshwater testate amoebae. Oxford: Oxford University Press. 222 p.
Oliverio A.M., Lahr D.J.G., Nguyen T., Katz L.A., 2014. Cryptic diversity within morphospecies of testate amoebae (Amoebozoa: Arcellinida) in New England bogs and fens // Protist. V. 165. P. 196‒207.
Oliverio A.M., Lahr D.J.G., Grant J., Katz L.A., 2015. Are microbes fundamentally different than macroorganisms? Convergence and a possible case of neutral phenotypic evolution in testate amoeba (Amoebozoa: Arcellinida) // Royal Society Open Science. V. 2. https://doi.org/10.1098/rsos.150414
Penard E., 1890. Études sur les Rhizopodes d’eau douce // Memoires de la Société de physique et d’histoire naturelle de Genève. V. 31. P. 1–230.
Penard E., 1905. Sarcodinés // Catalogue des Invertébrés de la Suisse. Volume I. Genève: Musée d’Histoire Naturelle de Genève. P. 1‒164.
Qin Y., Man B., Kosakyan A., Lara E., Gu Y., Wang H., Mitchell E.A.D., 2016. Nebela jiuhuensis nov. sp. (Amoebozoa; Arcellinida; Hyalospheniidae): a new member of the Nebela saccifera‒equicalceus‒ansata group described from Sphagnum peatlands in south-central China // Journal of Eukaryotic Microbiology. V. 63. P. 558‒566.
Reukauf E., 1912. Zur Encystierung von Euglypha alveolata // Zoologischer Anzeiger. V. 39. P. 372‒375.
Schönborn W., 1992. Adaptive polymorphisam in soil-inhabiting testate amoebae (Rhizopoda): its importance for delimitation and evolution of asexual species // Archiv für Protistenkunde. V. 142. P. 139‒155.
Schönborn W., Peschke T., 1988. Biometric studies on species, races, ecophenotypes and individual variations of soil-inhabiting Testacea (Protozoa, Rhizopoda), including Trigonopyxis minuta n. sp. and Corythion asperulum n. sp. // Archiv für Protistenkunde. V. 136. P. 345‒ 363.
Singer D., Kosakyan A., Pillonel A., Mitchell E.A.D., Lara E., 2015. Eight species in the Nebela collaris complex: Nebela gimlii (Arcellinida, Hyalospheniidae), a new species described from a Swiss raised bog // European Journal of Protistology. V. 51. P. 79–85.
Singer D., Kosakyan A., Seppey C.V.W., Pillonel A., Fernández L.D., Fontaneto D., et al., 2018. Environmental filtering and phylogenetic clustering correlate with the distribution patterns of cryptic protist species // Ecology. V. 99. P. 904‒914.
Smith H.G., Bobrov A., Lara E., 2007. Diversity and biogeography of testate amoebae // Protist diversity and geographical distribution. Foissner W., Hawksworth D.L. (Eds.). Dordrecht: Springer. P. 95‒109.
Št$\overset{\lower0.5em\hbox{$\smash{\scriptscriptstyle\smile}$}}{e} $pánek M., 1963. Die Rhizopoden aus Katanga (Kongo-Afrika) // Annales du Musée royal de l’Afrique centrale (Tervuren). V. 117. P. 9‒91.
Todorov M., 2002. Morphology, biometry and ecology of Nebela bigibbosa Penard, 1890 (Protozoa: Rhizopoda) // Acta Protozoologica. V. 41. P. 239–244.
Todorov M., 2010. Nebela golemanskyi sp. nov., a new sphagnicolous testate amoeba from Bulgaria (Amoebozoa: Arcellinida, Nebelidae) // Acta Protozoologica. V. 49. P. 37‒43.
Todorov M., Bankov N., 2019. An atlas of Sphagnum-dwelling testate amoebae in Bulgaria. Sofia: Pensoft. 286 p.
Todorov M., Bankov N., Ganeva A., 2018. Longinebela ampulla sp. n. (Arcellinida: Hyalospheniidae), a new testate amoeba from Sphagnum peatlands in Bulgaria // Acta Zoologica Bulgarica. V. 70. P. 285‒292.
Todorov M., Golemansky V., Meisterfeld R., 2010. Is Difflugia nebeloides (Amoebozoa: Arcellinida) really a Difflugia? Re-description and new combination // Acta Zoologica Bulgarica. V. 62. P. 13‒20.
Török J.K., 2001. Fine structure and biometric characterization of the shell in the rare testacean species Hyalosphenia punctata Penard (Protozoa: Testacealobosia) // Acta Protozoologica. V. 40. P. 291–296.
Van Breemen N., 1995. How Sphagnum bogs down other plants // Trends in Ecology & Evolution. V. 10. P. 270‒ 275.
Van Oye P., 1941. Die Rhizopoden des Sphagnetums bei Krisuvik auf Island // Biologisch Jaarboek (Dodonaea). V. 8. P. 284‒305.
Van Oye P., 1956. Rhizopoda Venezuelas. Mit bezonderer Berüchsichtigung ihrer Biogeographie // Ergebnisse der deutschen lomnologischen Venezuela-Expedition 1952. Volume I. Berlin: Deutscher Verlag der Wissenschaften. P. 329‒360.
Wailes G.H., 1912. Freshwater Rhizopoda and Heliozoa from the states of New York, New Yersey, and Georgia, U.S.A.; with supplemental notes on Seychelles species // Zoological Journal of the Linnean Society. V. 32. P. 121‒161.
Wanner M., 1999. A review on the variability on testate amoebae: methodological approaches, environmental influences and taxonomical implications // Acta Protozoologica. V. 38. P. 15‒29.
Weber H., 1965. Über die Paarung der Gamonten und den Kerndualismus der Foraminifere Metarotaliella parva Grell // Archiv für Protistenkunde. V. 108. P. 217‒270.
Yu Z., Zhang W., Liu L., Yang J., 2014. Evidence for two different morphotypes of Difflugia tuberspinifera from China // European Journal of Protistology. V. 50. P. 205‒ 211.
Zapata J., Fernández L., 2008. Morphology and morphometry of Apodera vas (Certes, 1889) (Protozoa: Testacea) from two peatlands in southern Chile // Acta Protozoologica. V. 47. P. 389–395.
Zapata J., Yáñez M., Rudolph E., 2008. Tecamebianos (Protozoa: Rhizopoda) de una turbera del Parque Nacional Puyehue (40°45′ S; 72°19′ W), Chile // Gayana. V. 72. P. 9‒17.
Дополнительные материалы отсутствуют.
Инструменты
Зоологический журнал


